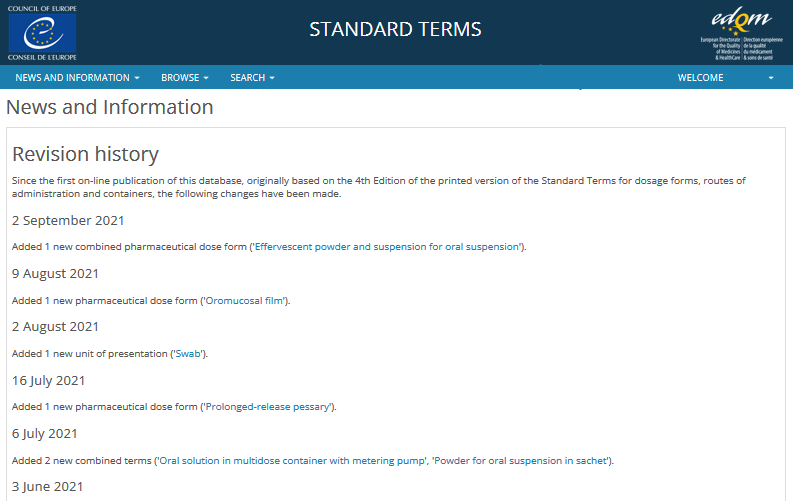
PDF) How Granular Can a Dose Form Be Described? Considering EDQM Standard Terms for a Global Terminology
European Directorate for the Quality of Medicines and Healthcare - European Directorate for the Quality of Medicines & HealthCare
Reminder: Use of EMA SPOR/OMS ORG_ID and LOC_ID mandatory for any CEP applications - European Directorate for the Quality of Medicines & HealthCare

Ph. Eur. Reference Standards: Orders and Catalogue - European Directorate for the Quality of Medicines & HealthCare
2024 EDQM virtual training programme: Ph. Eur. texts related to biologicals and microbiology chapters - European Directorate for the Quality of Medicines & HealthCare

European Directorate for the Quality of Medicines and Healthcare - European Directorate for the Quality of Medicines & HealthCare

PDF) How Granular Can a Dose Form Be Described? Considering EDQM Standard Terms for a Global Terminology
CEP 2.0: List of authorities and organisations which have access to assessment and/or inspection reports. - European Directorate for the Quality of Medicines & HealthCare

10 years of co-operation between the European Commission and the EDQM/Council of Europe in the field of blood: key achievements in standard setting - European Directorate for the Quality of Medicines &