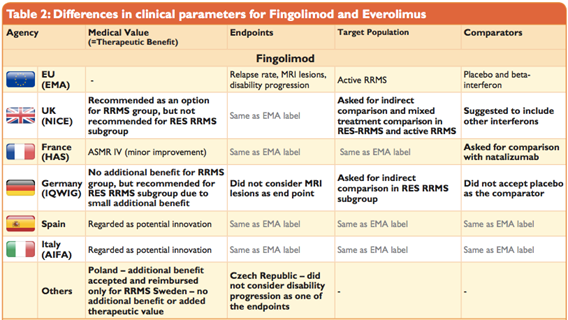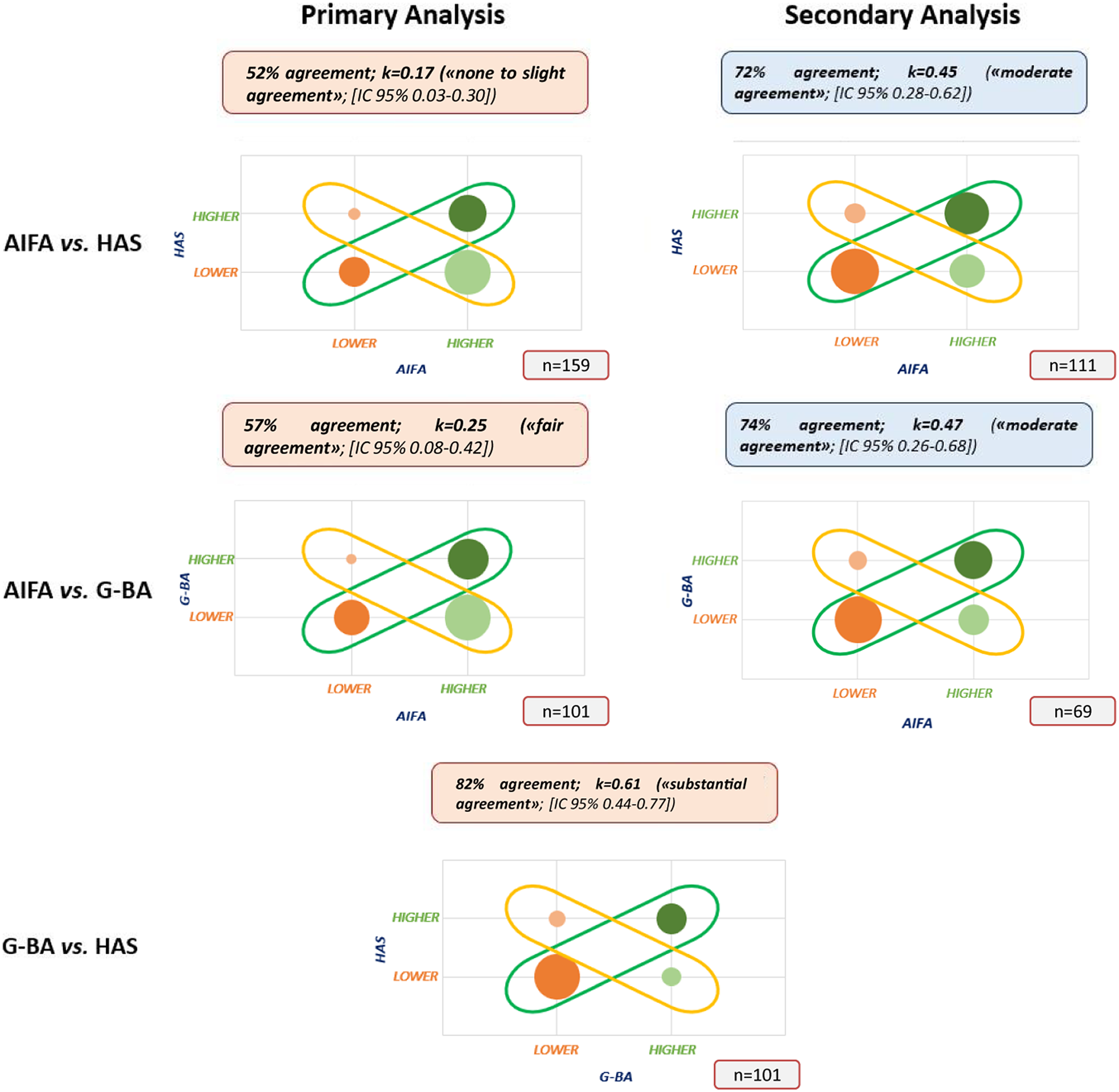
Do France, Germany, and Italy agree on the added therapeutic value of medicines? | International Journal of Technology Assessment in Health Care | Cambridge Core
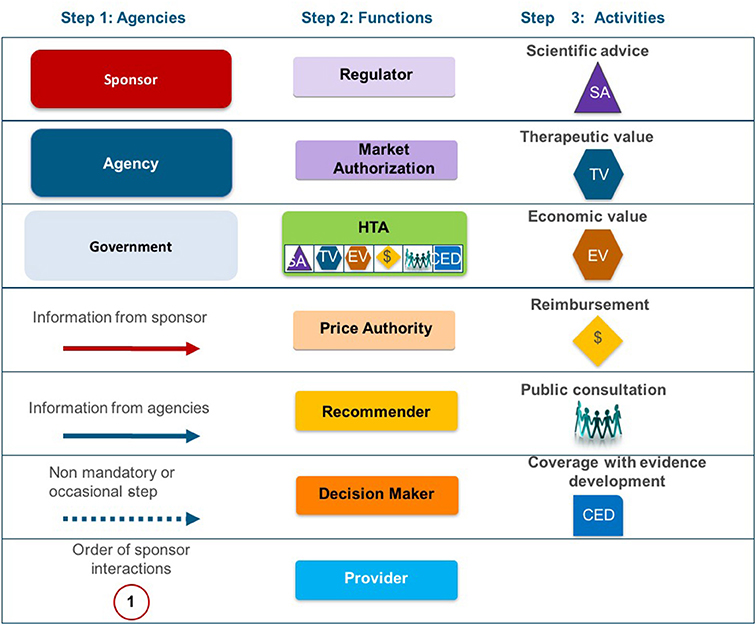
Frontiers | A Comparison of Reimbursement Recommendations by European HTA Agencies: Is There Opportunity for Further Alignment?

PDF) Weighing of Evidence by Health Technology Assessment Bodies: Retrospective Study of Reimbursement Recommendations for Conditionally Approved Drugs | Lourens T Bloem - Academia.edu

How can a joint European health technology assessment provide an 'additional benefit' over the current standard of national assessments? : Insights generated from a multi-stakeholder survey in hematology/oncology. - Abstract - Europe

Figure 5 from Health Technology Assessment (HTA) Case Studies: Factors Influencing Divergent HTA Reimbursement Recommendations in Australia, Canada, England, and Scotland. | Semantic Scholar

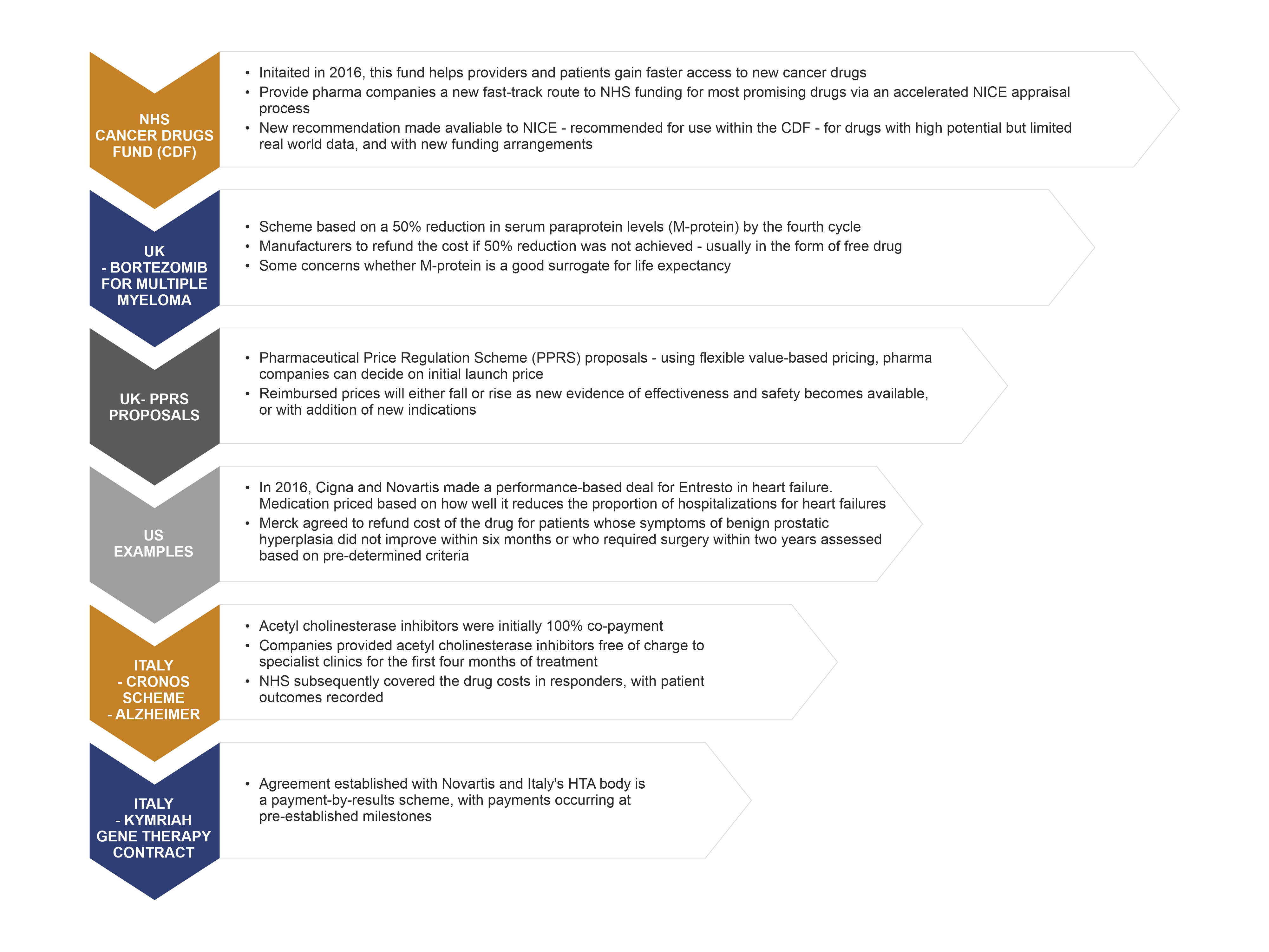
Europe's patchwork of evidence requirements is an important factor in delayed patient access - Consultancy in Healthcare and Life Sciences | Vintura Consultancy
Early engagement with Health Technology Assessment authorities will accelerate product launch and improve chances for reimbursement

Health technology assessment of medical devices: a survey of non-European union agencies. - Abstract - Europe PMC

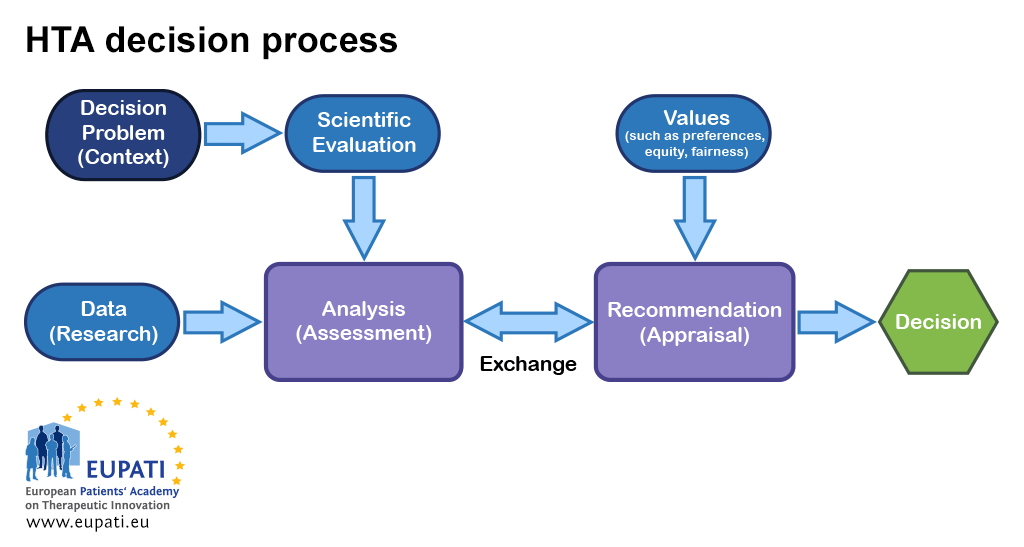
Frontiers | Towards a European harmonization of health technology assessment recommendations executive paper of European regulatory conference focused on the EU commission proposal to harmonize HTA

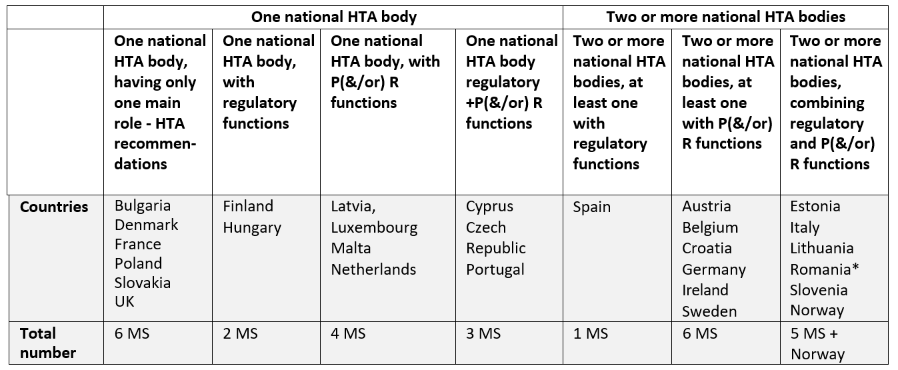
Similarities and Differences in Health Technology Assessment Systems and Implications for Coverage Decisions: Evidence from 32 Countries | PharmacoEconomics - Open