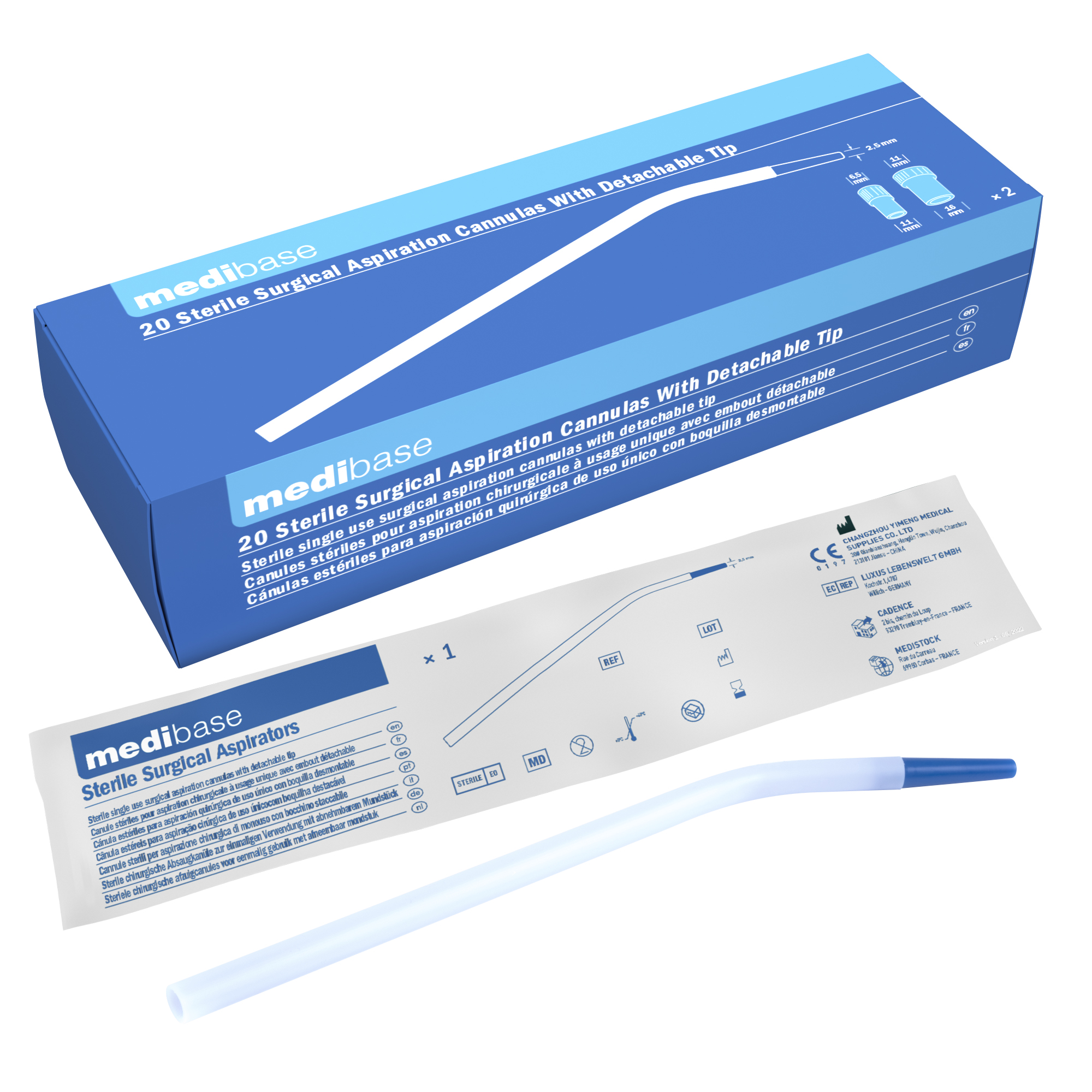
Organismos Notificados: TÜV Rheinland LGA Products ( @tuv_es ) NB num. 0197 nuevo ON con IVDR. Enhorabuena!!!

Which EU Notified Bodies Have Been “Designated” Under the MDR 2017/745 and IVDR 2017/746? – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog

Weldas Golden Brown™ Almohadilla de soldadura de cuero serraje vacuno (par), 15 cm L | Ropa protectora | Weldes

CE Marking a Medical Device under the EU MDR | Wellcome / EPSRC Centre for Interventional and Surgical Sciences - UCL – University College London

Mario Gabrielli Cossellu on LinkedIn: #notifiedbody #mdr #nando #ivdr #notifiedbodies #conformityassessment…
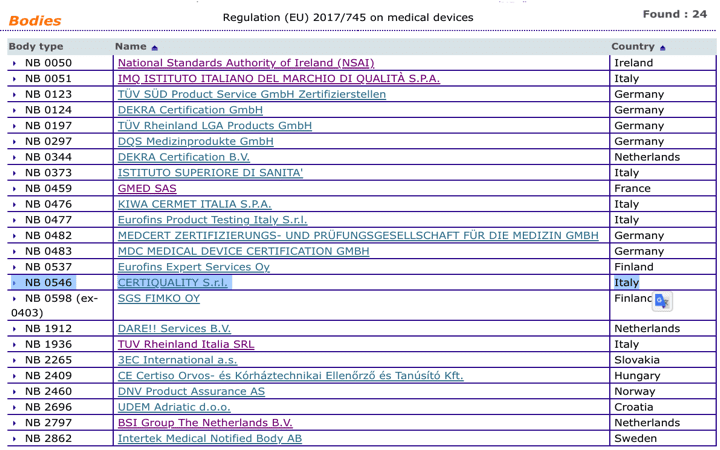
Organismos Notificados MDR (24): CERTIQUALITY (Italia) ON num. 0546 nuevo ON. Enhorabuena!!! | Red de Tecnologías Sanitarias y Productos Sanitarios

CGM accuracy: Contrasting CE marking with the governmental controls of the USA (FDA) and Australia (TGA): A narrative review - Pemberton - 2023 - Diabetes, Obesity and Metabolism - Wiley Online Library
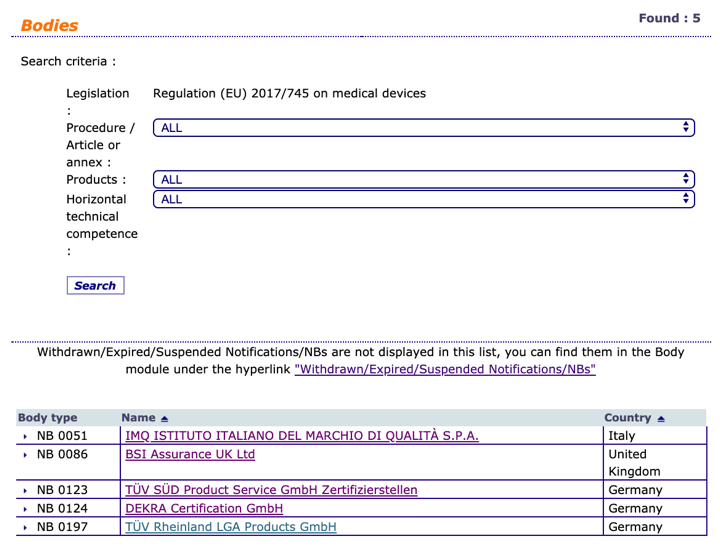
Organismos Notificados: TUV Rheinland LGA ( ON num. 0197 ) @TUV_es @TUVRheinlandNA nuevo Organismo Notificado con el reglamento MDR – Enhorabuena!! | Red de Tecnologías Sanitarias y Productos Sanitarios